The primary purpose of mounting metallographic samples is for convenience in handling samples of difficult shapes or sizes during the subsequent steps of metallographic preparation and examination. A secondary purpose is to protect and preserve extreme edges or surface defects during metallographic preparation. The method of mounting should in no way be injurious to the microstructure of the specimen. Pressure and heat are the most likely sources of injurious effects.
Phenolic plastics were introduced to metallography in 1928 for encapsulating specimens by hot compression mounting. Prior to that time, samples were prepared unmounted, or mounted in flours of sulfur, wax or low-melting point alloys, such as Wood’s metal. These “mounting compounds” were not without their problems. Introduction of polymers was a big improvement over these methods. Subsequently, many polymers were evaluated for use as mounting compounds, as they were introduced to the market. Development of castable resins in the 1950’s added new resins to the metallographer’s tool chest. The simplicity of mounting without using a press, and their low curing temperatures, made castable resins an attractive alternative.
Clamp Mounting
Clamps have been used for many years to mount cross sections of thin sheet specimens. Several samples can be clamped conveniently in sandwich form making this a quick, convenient method for mounting thin sheet specimens.
When done properly, edge retention is excellent, and seepage of fluids from crevices between specimens does not occur. The outer clamp edges should be beveled to minimize damage to polishing cloths. If clamps are improperly used so that gaps exist be- tween specimens, fluids and abrasives can become entrapped and will seep out obscuring edges and can cause cross contamination. This problem can be minimized by proper tightening of clamps, by use of plastic spacers between specimens, or by coating specimen surfaces with epoxy before tightening.
Compression Mounting
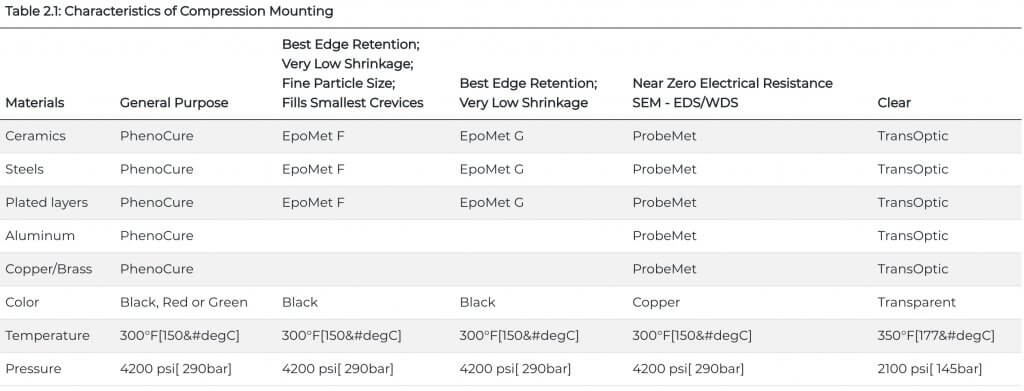
The most common metallographic mounting method uses pressure and heat to encapsulate the sample with a thermosetting or thermoplastic mounting material. Common thermosetting compounds include phenolic (PhenoCure), diallyl phthalate and epoxy (EpoMet) while methyl methacrylate (TransOptic) is the most commonly used thermoplastic mounting compounds. Table 2.1 lists the characteristics of the compression mounting compounds.
Both thermosetting and thermoplastic materials require heat and pressure during the molding cycle; but, after curing, mounts made of thermoplastic compounds must be cooled under pressure to at least 158°F [70°C] while mounts made of thermosetting materials may be ejected from the mold at the maximum molding temperature. However, cooling thermosetting compounds under pressure to near ambient temperature before ejection will significantly reduce shrinkage gap formation.
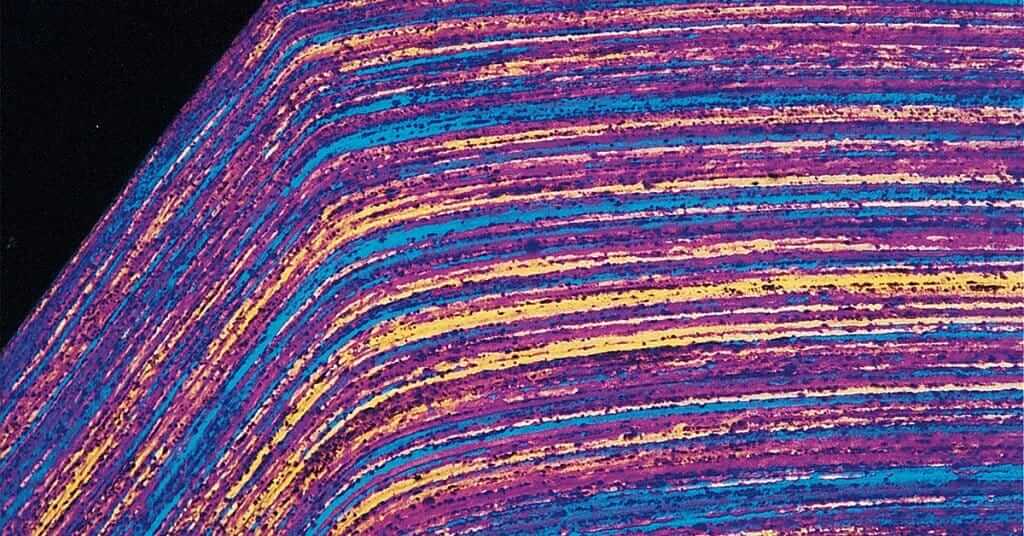
Never rapidly cool a thermosetting mount with water after hot ejection from the molding temperature. This causes the metal to pull away from the mounting compound producing shrinkage gaps that promote poor edge retention, see Figure 2.1, because of the different rates of thermal contraction.
 EpoMet, a thermosetting epoxy, provides the best edge retention, Figure 2.2, of these compounds and is virtually unaffected by hot or boiling etchants while phenolics are badly damaged.
EpoMet, a thermosetting epoxy, provides the best edge retention, Figure 2.2, of these compounds and is virtually unaffected by hot or boiling etchants while phenolics are badly damaged.
Mounting presses vary from simple laboratory jacks with a heater and mold assembly to full automated devices that include the SmartCool intelligent cooling system that ends the process as soon as the specimen is safe to handle, as shown in Figure 2.3.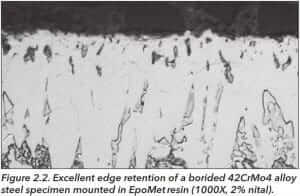 An advantage of compression mounting is production of a mount of a predictable, convenient size and shape. Further, considerable information can be engraved on the backside – this is always more difficult with unmounted specimens. Manual or hand, polishing is simplified, as the specimens are easy to hold.
An advantage of compression mounting is production of a mount of a predictable, convenient size and shape. Further, considerable information can be engraved on the backside – this is always more difficult with unmounted specimens. Manual or hand, polishing is simplified, as the specimens are easy to hold. Also, placing a number of mounted specimens in a holder for semi or fully automated grinding and polishing is easier with standard mounts than for unmounted specimens. Mounted specimens are easier on the grinding/polishing surfaces than unmounted specimens.
Also, placing a number of mounted specimens in a holder for semi or fully automated grinding and polishing is easier with standard mounts than for unmounted specimens. Mounted specimens are easier on the grinding/polishing surfaces than unmounted specimens.
Castable Mounting Systems
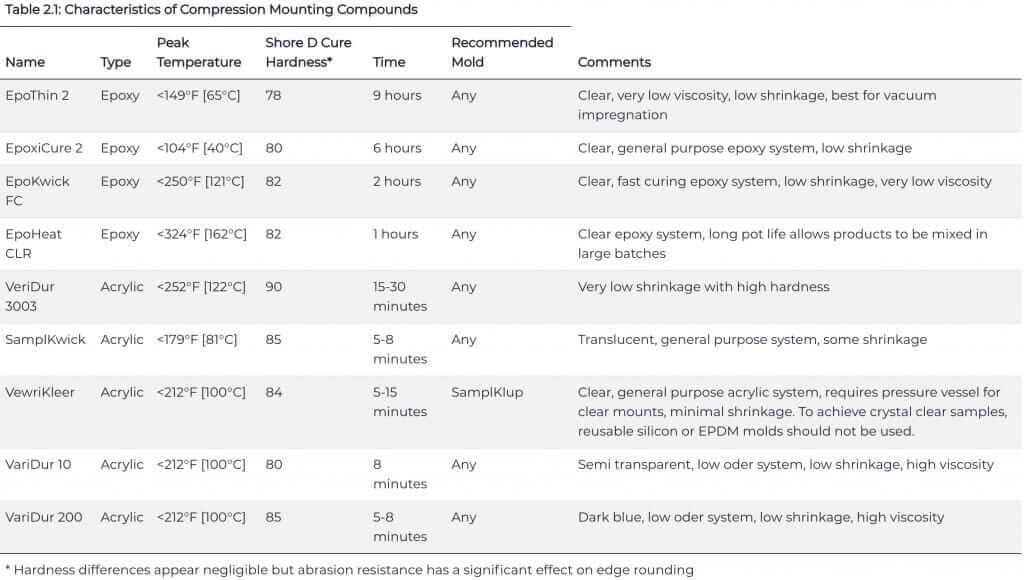
 Most castable mounting systems require neither pressure nor external heat and are recommended for mounting specimens that are sensitive to heat and/or pressure. Acrylic systems, such as VariDur and SamplKwick, are the most widely used castable systems due to their low cost and short curing time. However, shrinkage can be a problem with acrylics. Epoxy systems, although more expensive than acrylics, are commonly used because epoxy will physically adhere to specimens, have low shrinkage, and can be drawn into cracks and pores, particularly if a vacuum impregnation chamber, as shown in Figure 2.4, is employed and a low viscosity epoxy, such as EpoThin 2, is used.
Most castable mounting systems require neither pressure nor external heat and are recommended for mounting specimens that are sensitive to heat and/or pressure. Acrylic systems, such as VariDur and SamplKwick, are the most widely used castable systems due to their low cost and short curing time. However, shrinkage can be a problem with acrylics. Epoxy systems, although more expensive than acrylics, are commonly used because epoxy will physically adhere to specimens, have low shrinkage, and can be drawn into cracks and pores, particularly if a vacuum impregnation chamber, as shown in Figure 2.4, is employed and a low viscosity epoxy, such as EpoThin 2, is used.
Epoxies are very suitable for mounting fragile or friable specimens and corrosion or oxidation specimens. Dyes or fluorescent agents may be added to epoxies for the study of porous specimens such as thermal spray coated specimens. Most epoxies are cured at room temperature, and curing times can vary from 1 to 9 hours. Some can be cured at slightly elevated temperatures in less time, as long as the higher temperature does not adversely affect the specimen. Table 2.1 below lists the characteristics of castable systems.
Cast epoxy systems provide better edge retention than cast acrylic systems, mainly due to the better adhesion of epoxy to the specimen and their lower shrinkage. Acrylics usually do not bond to the specimen and a gap is sometimes formed between specimen and mount, with VariDur 3003 being the exception. When a shrinkage gap is present, edge retention is usually poor. To improve edge retention with castable mounts, the specimen can be plated with electroless nickel, or Flat Edge Filler particles can be added to the system. To obtain electrical conductivity, Conductive Filler particles can be added to the castable systems, though the viscosity increases.
When preparing castable resin mounts, particularly epoxy mounts by manual “hand” methods, the metallographer will observe that the surface tension between the mount and the working surface is much higher than with a compression mount. This can make holding the mount more challenging. If automated devices are used, the metallographer may hear “chatter,” or noise, during rough grinding due to the greater surface tension. The chatter can be reduced or stopped by changing to contra mode (head and platen rotate in opposite directions).
Acrylics, and some epoxies, do generate considerable heat during curing and this can be strongly influenced by the molding technique used. Nelson measured the exotherm produced by polymerizing an acrylic system using two procedures: a glass mold on a glass plate (insulative) and an aluminum mold on an aluminum plate (conductive).
Polymerization produced a maximum exotherm of 270°F [132°C] using the insulative approach but only 108°F [42°C] using the conductive approach. Note that 270°F [132°C] is not much less than the 302°F [150°C] temperature used in compression mounting. Nelson also measured the exotherm produced when an epoxy system was cured in a phenolic ring form placed on a pasteboard base. Although this was an insulative approach, the maximum temperature during polymerization was only 45°F [7°C], a vast improvement over the acrylics.
Nelson’s work applies to specific acrylic and epoxy resins molded upon specific conditions. While the epoxy that he used exhibited a low exotherm, this does not imply that all epoxy systems will exhibit such low exotherms in polymerization. Epoxy systems that cure in short time periods develop much higher exotherms, that can even exceed that of acrylic systems. In addition to the speed of curing of the epoxy system, other factors do influence the magnitude of the exotherm during polymerization. The larger the mass of epoxy in the mount, the faster it will set and the greater the exotherm. Indeed, very large mounts can generate enough heat to crack extensively. Heating the system makes it less viscous and speeds up curing, also generating more heat during polymerization. The mold material also can influence curing time and temperature.

For example, EpoxiCure 2 cures fastest in SamplKup plastic molds, slower in ring forms, and still slower in the reuseable EPDM mounting cups. Consequently, the exotherm will be greater when using the SamplKup type mold and lowest when using the EPDM mounting cups. All of these factors must be considered if the exotherm must be minimized.
Edge Preservation
 Edge preservation is a classic preparation problem and many “tricks” have been promoted (most pertaining to mounting, but some to grinding and polishing) to enhance edge flatness. These methods include the use of backup material in the mount, the application of coatings to the surfaces before mounting or the addition of a filler material to the mounting system. Plating of a compatible metal on the surface to be protected (electroless nickel has been widely used) is generally considered to be the most effective procedure. However, image contrast at an interface between a specimen and the electroless nickel may be inadequate for certain evaluations.
Edge preservation is a classic preparation problem and many “tricks” have been promoted (most pertaining to mounting, but some to grinding and polishing) to enhance edge flatness. These methods include the use of backup material in the mount, the application of coatings to the surfaces before mounting or the addition of a filler material to the mounting system. Plating of a compatible metal on the surface to be protected (electroless nickel has been widely used) is generally considered to be the most effective procedure. However, image contrast at an interface between a specimen and the electroless nickel may be inadequate for certain evaluations.
Figure 2.5 shows the surface of a specimen of 1215 free-machining steel that was salt bath nitrided. One specimen was plated with electroless nickel; both were mounted in EpoMet G. It is hard to tell where the nitrided layer stops for the plated specimen, Figure 2.5a, which exhibits poor image contrast between the nickel and the nitrided surface. This is not a problem for the non-plated specimen, Figure 2.5b.
 Introduction of new technology has greatly reduced edge preservation problems. Gaps that form between specimen and mount media are a major contributor to edge rounding, as shown in Figure 2.1. Staining at shrinkage gaps may also be a problem, as demonstrated in Figure 2.6.
Introduction of new technology has greatly reduced edge preservation problems. Gaps that form between specimen and mount media are a major contributor to edge rounding, as shown in Figure 2.1. Staining at shrinkage gaps may also be a problem, as demonstrated in Figure 2.6.
Use of semi-automatic and automatic grinding/polishing equipment, rather than manual preparation, increases surface flatness and edge retention. To achieve the best results, however, the position of the specimen holder, relative to the platen, must be adjusted so that the outer edge of the specimen holder rotates out over the edge of the surface on the platen during grinding and polishing, particularly for 8in [203mm] diameter platens. The use of harder, woven or non-woven, napless surfaces for polishing with diamond abrasives (rather than softer cloths such as canvas, billiard and felt) maintains flatness. Final polishing with low nap cloths for short times introduces very little rounding compared to use of higher nap, softer cloths.
These procedures will produce better edge retention with all thermosetting and thermoplastic mounting compounds. Nevertheless, there are still differences among the polymeric materials used for mounting. Thermosetting compounds provide better edge retention than thermoplastic compounds.
Of the thermosetting compounds, diallyl phthalate provides little improvement over the much less expensive phenolic compounds. The best results are obtained with EpoMet G or EpoMet F an epoxy based thermosetting compounds that contains a filler material. For comparison, Figure 2.7 shows micrographs of a nitrided 1215 specimen mounted in a phenolic (Figure 2.7a), and in methyl methacrylate (Figure 2.7b), at 1000X. These specimens were prepared in the same specimen holder as those shown in Figure 2.5, but neither displays acceptable edge retention at 1000X. Figure 2.8 shows examples of perfect edge retention, as also demonstrated in Figure 2.5.
For comparison, Figure 2.7 shows micrographs of a nitrided 1215 specimen mounted in a phenolic (Figure 2.7a), and in methyl methacrylate (Figure 2.7b), at 1000X. These specimens were prepared in the same specimen holder as those shown in Figure 2.5, but neither displays acceptable edge retention at 1000X. Figure 2.8 shows examples of perfect edge retention, as also demonstrated in Figure 2.5.
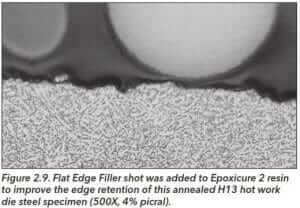
Very fine aluminum oxide spheres have been added to castable epoxy systems to improve edge retention, but this is really not a satisfactory solution as the particles are extremely hard (~2000 HV) and their grinding-polishing characteristics are incompatible with softer metals placed inside the mount. However, use of a soft ceramic shot (~775 HV) has grinding/polishing characteristics compatible with metallic specimens placed in the mount.
Figure 2.9 shows an example of edge retention with the Flat Edge Filler soft ceramic shot in an epoxy system.
 Following are general guidelines for obtaining the best possible edge retention. All of these factors contribute to the overall success, although some are more critical than others.
Following are general guidelines for obtaining the best possible edge retention. All of these factors contribute to the overall success, although some are more critical than others.
- Properly mounted specimens yield better edge retention than unmounted specimens, as rounding is difficult, if not impossible, to prevent at a free edge. Compression mounts yield better edge preservation than castable systems.
- Electrolytic or electroless Ni plating of the surface of interest provides excellent edge retention. If the compression mount is cooled too quickly after polymerization, the plating may be pulled away from the specimen leaving a gap. When this happens, the plating is ineffective for edge retention.
- Thermoplastic compression mounting compounds are less effective than thermosetting compounds. The best thermosetting compound for edge retention is EpoMet G, an epoxy-based resin containing a hard filler material.
- Never hot eject a thermosetting compound after polymerization and cool it quickly to ambient (e.g., by cooling it in water) as a gap will form between specimen and mount due to the differences in thermal contraction rates. Fully automated mounting presses cool the mounted specimen to near ambient temperature under pressure and this greatly minimizes gap formation due to shrinkage.
- Automated grinding-polishing equipment produces flatter specimens than manual preparation.
- Use the central force mode (defined later in the text) with an automated grinder-polisher as this method provides better flatness than single, or individual, pressure mode (defined later in the text).
- Orient the position of the smaller diameter specimen holder so that, as it rotates, its periphery slightly overlaps the periphery of the larger diameter platen.
- Use PSA-backed SiC grinding paper (if SiC is used), rather than plain backed paper on the platen and a peripheral holddown ring, and PSA-backed or magnetic polishing cloths rather than stretched cloths.
- Apex DGD and UltraPrep diamond grinding discs produce excellent flat surfaces for a wide variety of materials.
- Use “hard” napless surfaces for rough polishing (until the final polishing step), such as TexMet C, UltraPol or UltraPad cloths, and fine polishing, such as a TriDent cloth. Use a napless, or a low- to medium-nap cloth, depending upon the material being prepared, for the final step and keep the polishing time brief.
- Rigid grinding discs, such as the Apex Hercules H and S discs, produce excellent flatness and edge retention and should be used whenever possible.
Helpful Hints for Mounting
Castable resins are sensitive to shelf life. It is a good practice to date your containers when you get them.
For more information refer to our mounting equipment and the Buehler SumMet Guide.